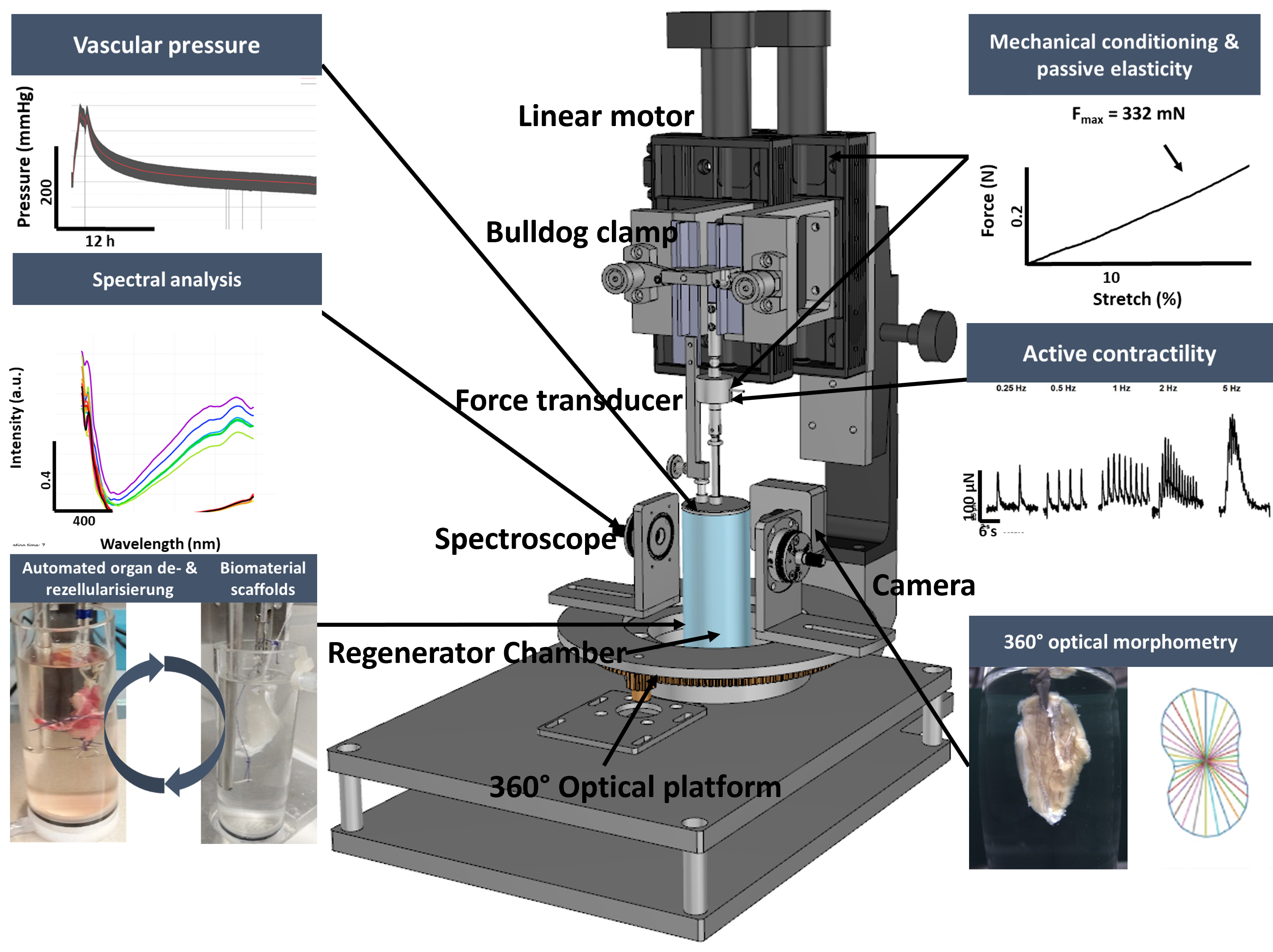
The TissueRegenerator is designed to set the foundation for future in-vitro organ regeneration through a concept we call Adaptive Tissue Engineering. This field is hallmarked by automated and adaptive technology relying on sophisticated process engineering, thorough sample and environmental monitoring (preferentially contactless) and smart process control strategies. Current progress in organ de- and recellularization largely relies on excessive staff and material resources, involved in a multi-weeks manual process, which, by nature, is variably error-prone. Moreover, even the process of creating cell-free organ scaffolds (decellularization) – the starting point for regenerative studies – suffers from destructive end-point quality assessments, e.g., fixation, slicing & staining (microscopy), or grinding (DNA quantification). This would also account for the recellularized organ and inevitably renders it unusable for further experimentation and obstructs clinical translation. Therefore, we engineered our patented TissueRegenerator and defined major tasks to succeed with an adaptive tissue engineering process for organ regeneration:
- replace destructive end-point assessments with contactless quality criteria
- provide decellularized matrix scaffolds of standardized quality
- eliminate ‚time‘ as the major determinant for successful de- and recellularization
- provide an adaptive and multi-modal environmental- and sample-monitoring unit
- rely on integrated sensor information to supervise cell injection, proliferation & well-being
- have biomimicking (mechanical & electrical stimulation) assisting the regeneration process
- measure tissue performance and function as it regenerates
- optimize the process based on the sensor information to achieve a status ‚like-native‘
 1/2
1/2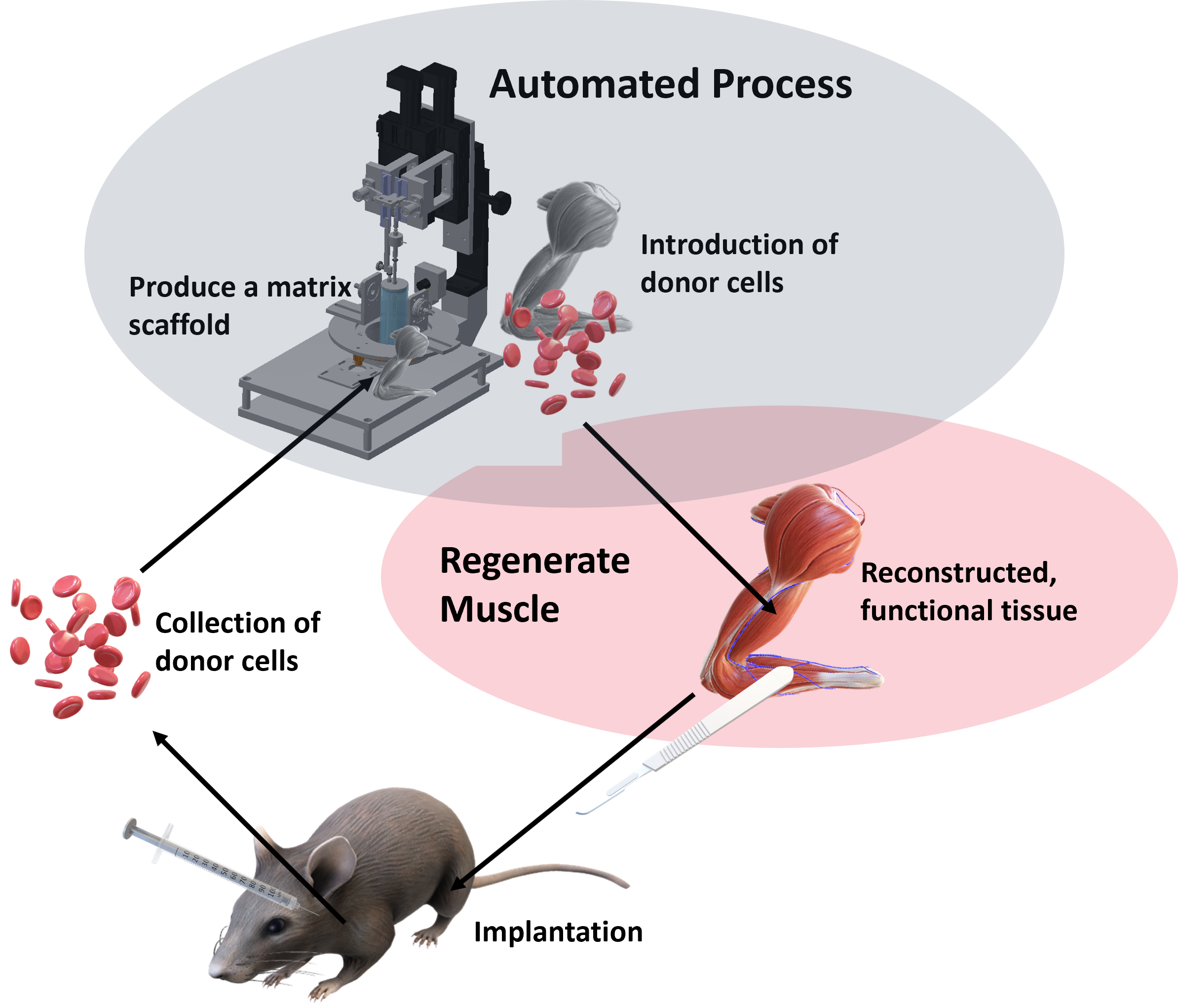 2/2
2/2References:
- Ritter P, Cai A, Reischl B, et al. (2022) MyoBio: Am Automated Bioreactor System Technology for Standardized Perfusion-Decellularization of Whole Skeletal Muscle. IEEE Transactions on Biomedical Engineering. 10.1109/TBME.2022.3142317.
- DE-Patent 10 2021 211 875. Bioreaktor und Verfahren zum Betrieb eines solchen. DPMA.
- PCT Anmeldung: PCT/EP 2022/076504
